
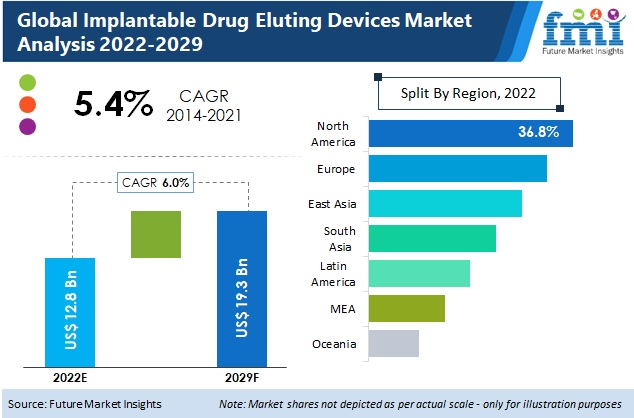
Implantable Drug-Eluting Devices Market is expected to reach a valuation of around US$ 12.8 Billion as of 2022
[326 Pages Report] Implantable drug-eluting devices are playing a key role in the management and treatment of various chronic disorders, particularly cardiovascular diseases. Advancements in micro and nanotechnology are aiding key manufacturers to design novel implantable drug-eluting devices.
Moreover, controlled and site-specific attributes of implantable drug-eluting devices, further complemented by compatibility with the targeted drug delivery, are relieving the burden of complex dosing schedule and systemic toxicity of conventional drug delivery system. The global implantable drug-eluting devices market is projected to cross the US$ 10 Bn mark towards the end of 2022. Market leaders in implantable drug-eluting devices landscape are maintaining their focus on refining manufacturing processes.
Growth of implantable drug-eluting devices market can be prominently attributed to the associated cost-effective technology and novel medication administration techniques. Fewer side effects while these devices are utilized in the management of chronic diseases as well as in contraception have been the strongest attribute accounting for a steadily expanding market for implantable drug-eluting devices.
Remote-Controlled Chips and Programmable Devices – Future of Targeted Drug Delivery
Increasing clinical trials and research & development of remote-controlled and programmable drug-eluting devices
Massachusetts Institute of Technology (MIT) researchers have developed a remote-controlled contraceptive hormone (levonorgestrel) delivery chip implant, which can turn the device on and off.
Implantable drug-eluting devices allow minimum effective doses and support oral opioid weaning
Implantable drug-eluting devices such as SynchroMed II intrathecal drug delivery system with controlled workflow system, plays an effective role in reducing or eliminate the use of oral opioids.
Antiretroviral (ARV) drug implant is expected to emerge as a promising approach for HIV treatment and prevention
In July 2022, Merck & Co. started the development of slow-release antiretroviral (ARV) drug implant as a promising approach for HIV treatment.
To remain ahead of your competitors, request for a sample – https://www.futuremarketinsights.com/reports/sample/rep-gb-10671
Know More About Implantable Drug-Eluting Devices Market Report
The implantable drug-eluting devices market, a new study from Future Market Insights, opines on the evolution of the implantable drug-eluting devices market from 2014 – 2021 and presents demand projections from 2022 – 2029 on basis of product type (implantable drug infusion pumps, intraocular drug-eluting devices, contraceptive drug-eluting devices, buprenorphine implant and drug-eluting stents), technology (active drug-eluting devices and passive drug-eluting devices) applications (diabetes macular edema, birth control/contraception, opioid addiction, cardiovascular, ophthalmology, oncology, pain management, others) implantation type (intravaginal, intravascular, intraocular, intrathecal, subcutaneous, intrauterine) end users (hospitals, ambulatory surgical centers, optical care centers, cardiac surgery centers, office-based setting and others) in seven prominent regions.
Medical-Pharma Collaborating to Balance Demand and Supply
A comprehensive view of the implantable drug-eluting devices market has led our analysts to conclude that, the market is expected to grow with an impressive rate. This market growth is mainly attributed due to increasing innovations and developments in implantable drug-eluting devices for the management and treatment of breast cancer, schizophrenia, Parkinson’s disease, and photosensitivity diseases. Besides, FMI analyst suggests that markets in North America and Europe are expected to hold major value shares in global implantable drug-eluting devices landscape. To cater to rising demand for implantable drug-eluting devices, medical device companies are collaborating with pharmaceutical companies for the development of efficient drug delivery devices.
Increasing use of syringe-based subcutaneous implantable drug-eluting devices is anticipated to minimize the number of invasive surgical procedures. Introduction of implantable drug-eluting devices or rare conditions such as central precocious puberty (CPP) will emerge as an important factor shaping the market for implantable drug-eluting devices.
Key Market Segments Covered in Implantable Drug Eluting Devices Industry Research
By Product:
- Implantable Drug Infusion Pumps
- Intraocular Drug Eluting Devices
- Contraceptive Drug Eluting Devices
- Buprenorphine Implant
- Drug Eluting Stents
- Others
By Technology:
- Active Drug Eluting Device
- Passive Drug Eluting Device
- Biodegradable Implants
- Non-Biodegradable Implants
By Application:
- Diabetic Macular Edema
- Birth Control/Contraception
- Opioid Addiction
- Cardiovascular
- Ophthalmology
- Oncology
- Pain Management
Get a Tailored Made Report to Match Your requirements, Ask from Market Research Expert – https://www.futuremarketinsights.com/ask-question/rep-gb-10671
About Future Market Insights (FMI)
Future Market Insights (FMI) is a leading provider of market intelligence and consulting services, serving clients in over 150 countries. FMI is headquartered in Dubai, and has delivery centers in the UK, U.S. and India. FMI’s latest market research reports and industry analysis help businesses navigate challenges and make critical decisions with confidence and clarity amidst breakneck competition. Our customized and syndicated market research reports deliver actionable insights that drive sustainable growth. A team of expert-led analysts at FMI continuously tracks emerging trends and events in a broad range of industries to ensure that our clients prepare for the evolving needs of their consumers.
Contact:
Future Market Insights,
1602-6 Jumeirah Bay X2 Tower,
Plot No: JLT-PH2-X2A,
Jumeirah Lakes Towers, Dubai,
United Arab Emirates
For Sales Enquiries: sales@futuremarketinsights.com
For Media Enquiries: press@futuremarketinsights.com
Website: https://www.futuremarketinsights.com/
Editor Details
-
Company:
- MARKITWIRED
- Website:
