
ADOCIA Announces Exceptional Weight Loss for Obese People with Type 1 Diabetes using M1Pram in a post-hoc analysis
- A weight loss of 5.5kg measured at 16 weeks of treatment in people with type 1 diabetes and a body mass index greater than 30kg/m2
- Glycemic control efficacy comparable to that obtained with the commercial reference insulin Humalog® (Eli Lilly)
- These results were presented at the 58th European Association for the Study of Diabetes (EASD) Annual Meeting held in Stockholm from September 19 to 23, 2022
LYON, France--(BUSINESS WIRE)--Regulatory News:

Adocia (Euronext Paris: FR0011184241 – ADOC), a clinical-stage biopharmaceutical company focused on the research and development of innovative therapeutic solutions for the treatment of diabetes and other metabolic diseases, announces outstanding additional results from its Phase 2 study with M1Pram in obese people with type 1 diabetes.
M1PRAM, ADDRESSING THE UNMET MEDICAL NEED OF OBESITY IN PEOPLE WITH TYPE 1 DIABETES
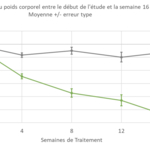
Post-hoc analyses revealed the greater efficacy of M1Pram in a subpopulation of obese patients with a Body Mass Index (BMI) greater than 30kg/m2. Weight loss in the M1Pram arm was -5.56kg versus -0.57 kg (p=0.03) in the Humalog arm at week 16, and weight loss had not plateaued by the end of the study.
The satisfaction questionnaire clearly demonstrated better appetite control with M1Pram for 82.4% of patients (versus 43.2% with Humalog).
"Given that more and more people with type 1 diabetes are overweight or obese, the availability of M1Pram allows these patients to maintain glycemic control while losing weight, an important outcome”, declared Jay S. Skyler, Professor of Medicine, University of Miami Leonard M. Miller School of Medicine.
As a reminder, the CT041 Phase 2 clinical trial was comparing M1Pram to insulin lispro (Humalog®, Eli Lilly). The positive results on the total population were communicated on June 21, 20221. The study included people with type 1 diabetes and a body mass index greater than 25kg/m2 (overweight and obese people). The primary endpoint of the trial was met with a significant weight loss of M1Pram vs Humalog over 4 months of -2.13kg (p=0.0045).
WHILE REDUCING WEIGHT, M1PRAM OFFERS GLYCEMIC CONTROL AS GOOD AS GOLD STANDARD MEALTIME INSULIN
M1Pram demonstrated to be equivalent to Humalog in controlling blood glucose, as safe in terms of risk of hypoglycemia and as convenient in terms of use.
- Both treatments maintain HbA1c levels and Time-In-Range in patients with a mean HbA1c level of 7.4% at baseline
- The number and severity of hypoglycemic events are similar in both treatment arms
- This coformulation is injected at mealtime by single injection
In addition, M1Pram reduced the daily dose of prandial insulin by 21% in the general population of the study.
M1Pram had an overall good safety profile. The difference in total adverse events of M1Pram versus Humalog (76 vs. 38) was mainly due to gastrointestinal side effects as documented in the pramlintide literature.
OBESITY, A MAJOR BURDEN IN PEOPLE WITH TYPE 1 DIABETES AND AN UNMET MEDICAL NEED
Nine million people in the world currently suffer from type 1 diabetes and this figure will double in the coming years2. 65%3 of them are overweight (BMI>25kg/m2) and about 37%4 are obese (BMI>30kg/m2) probably mainly due to the anabolic effect of insulin used daily to regulate diabetes.
Moreover, emerging evidence suggests that obesity contributes to insulin resistance, dyslipidemia, and cardiometabolic complications in type 1 diabetes5.
To date, pramlintide is the only product reducing weight that is approved by the FDA as an adjunct to insulin for people with type 1 diabetes.
M1PRAM TO REPLACE MEALTIME INSULINS FOR OBESE PEOPLE WITH DIABETES
M1Pram combines M1 insulin and pramlintide in a regular insulin pen. M1Pram coformulation is patented by Adocia until 2038.
Pramlintide is an amylin analog that is FDA approved as an adjunct to insulin in type 1 and type 2 diabetes. Pramlintide has demonstrated having significant effects in improving glycemic control, weight loss in overweight patients and well-being. Despite its clinical benefits, pramlintide has never been largely used by patients because it requires 3 additional injections on top of insulin daily injections, these two hormones normally being incompatible in one formulation. Based on 15-years of experience in protein formulation and diabetes, Adocia has successfully formulated pramlintide and insulin together in one single pen.
“At the EASD Annual Meeting, the uniqueness of M1Pram, mealtime insulin allowing significant weight loss, attracted the interest of several companies involved in the treatment of diabetes. Our goal now is to establish a strategic partnership to continue the development of M1Pram in obese people with type 1 Diabetes and extend it to obese people with type 2”, declared Olivier Soula, Deputy CEO of Adocia.
About Adocia
Adocia is a biotechnology company specializing in the discovery and development of therapeutic solutions in the field of metabolic diseases, primarily diabetes and obesity. The company has a broad portfolio of drug candidates based on three proprietary technology platforms:
1) The BioChaperone® technology for the development of new generation insulins and products combining insulins with other classes of hormones; 2) AdOral®, an oral peptide delivery technology; 3) AdoShell®, an immunoprotective biomaterial for cell transplantation with a first application in pancreatic cells transplantation for patients with "brittle" diabetes.
Adocia holds more than 25 patent families.
Based in Lyon, the company has approximately 105 employees. Adocia is listed on the EuronextTM Paris market (Euronext: ADOC; ISIN: FR0011184241).
Disclaimer
This press release contains certain forward-looking statements concerning Adocia and its business. Such forward-looking statements are based on assumptions that Adocia considers as being reasonable. However, there can be no guarantee that the estimates contained in such forward-looking statements will be achieved, as such estimates are subject to numerous risks including those which are set forth in the “Risk Factors” section of the universal registration document that was filed with the French Autorité des marchés financiers on April 21, 2022 (a copy of which is available at www.adocia.com, in particular uncertainties that are linked to research and development, future clinical data, analyses, and the evolution of the economic context, the financial markets and the markets in which Adocia operates.
The forward-looking statements contained in this press release are also subject to risks not yet known to Adocia or not considered as material by Adocia as of this day. The occurrence of all or part of such risks could cause that actual results, financial conditions, performances, or achievements of Adocia be materially different from those mentioned in the forward-looking statements.
1https://www.adocia.com/wp-content/uploads/2022/06/PR-Adocia-M1Pram-Topline-Results-ENG.pdf
2 Type 1 Diabetes Index https://www.t1dindex.org/
3 Conway B, Miller RG, Costacou T, Fried L, Kelsey S, Evans RW, Orchard TJ. Temporal patterns in overweight and obesity in Type 1 diabetes. Diabet Med. 2010 Apr;27(4):398-404. doi: 10.1111/j.1464-5491.2010.02956.x. PMID: 20536510; PMCID: PMC3129711.
4 Amelia S Wallace, Alex R Chang, Jung-Im Shin, Jodie Reider, Justin B Echouffo-Tcheugui, Morgan E Grams, Elizabeth Selvin, Obesity and Chronic Kidney Disease in US Adults With Type 1 and Type 2 Diabetes Mellitus, The Journal of Clinical Endocrinology & Metabolism, Volume 107, Issue 5, May 2022, Pages 1247–1256, https://doi.org/10.1210/clinem/dgab927
5Corbin KD, Driscoll KA, Pratley RE, Smith SR, Maahs DM, Mayer-Davis EJ; Advancing Care for Type 1 Diabetes and Obesity Network (ACT1ON). Obesity in Type 1 Diabetes: Pathophysiology, Clinical Impact, and Mechanisms. Endocr Rev. 2018 Oct 1;39(5):629-663. doi: 10.1210/er.2017-00191. PMID: 30060120.
Contacts
Adocia
Gérard Soula
CEO
contactinvestisseurs@adocia.com
Tel : +33 (0)4 72 610 610
www.adocia.com
Ulysse Communication
Adocia Press and Investors Relations
Pierre-Louis Germain
Margaux Puech Pays d’Alissac
Bruno Arabian
adocia@ulysse-communication.com
+ 33 (0)6 64 79 97 51
Editor Details
-
Company:
- Businesswire
